International relation
International Cooperation
- Overall Cooperation

- Bilateral Cooperation
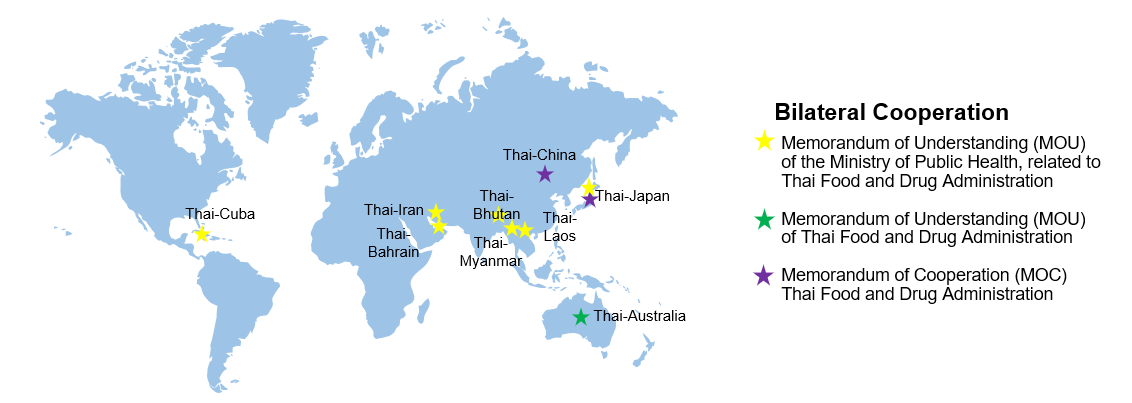 .
.
Currently, the Food and Drug Administration has signed Memorandums of Understanding (MOUs) and Memorandums of Cooperation (MOC) with 3 other countries to develop bilateral cooperations regarding health products, being:
1. Thailand - Australia
Memorandum of Understanding between the Food and Drug Administration of the Kingdom of Thailand and the Therapeutic Goods Administration of the Commonwealth of Australia on Therapeutic Products was signed by Adjunct Professor John Skerritt, Deputy Secretary, Health Products Regulation Group, Department of Health of Australia, Australia on 13th June 2021 and by the Secretary-General of the Food and Drug Administration, Thailand on 18th February 2021.
The MOU has entered into force since the day of its signature and covers the exchange of confidential information regarding therapeutic products which consist of drugs, medical devices, narcotic substances and herbal products, cooperative activities, as well as exchange of personnel, collaborative researches in relation to the quality, safety or efficacy of therapeutic products, or joint meetings in relation to rules and regulations regarding therapeutic products, which would benefit the capacity building of human resources of the FDA and strengthen the regulation of therapeutic products in Thailand.
2. Thailand - People’s Republic of China
Memorandum of Cooperation between the Food and Drug Administration of the Kingdom of Thailand and the State Food and Drug Administration of the People’s Republic of China was signed on 18th January 2007, and has entered into force since the day of its signature. The MOC covers cooperations relating to the Food and Drug Administration as follows: (1) exchange of information and promotion of cooperation regarding rules, regulations, standards and registration systems in relation to drugs, which include Chinese traditional medicinal drugs; (2) exploration of the possibility of cooperation on clinical research on
anti-HIV drugs in Thailand and China; (3) cooperation on rehabilitative drugs for narcotic addicts; and (4) exchange of information and promotion of cooperation on food safety.
3. Thailand - Japan
Memorandum of Cooperation between the Food and Drug Administration, Ministry of Public Health, the Kingdom of Thailand and the Ministry of Health, Labour and Welfare, Japan on Medical Products was signed on 9th April 2018 in Tokyo, Japan.
The MOC will remain in effect for 5 years and will be automatically extended every 5 years, and covers cooperations relating to the Food and Drug Administration as follows: the holding of annual meeting to discuss important issues concerning laws and regulations in relation to medical products in Thailand and Japan, exchange of best practices, and revision of rules and regulations in relation to medical products for the purpose of legal harmonization where it is mutually beneficial.
- Multilateral Cooperations
1. International Cooperation on Health Products under ASEAN
1.1 Agreement on the ASEAN Harmonized Cosmetic Regulatory Scheme (AHCRs)
ASEAN Economic Ministers of all 10 ASEAN Member States, being Brunei Darussalam, Cambodia, Indonesia, Laos, Malaysia, Myanmar, Philippines, Singapore, Thailand, and Vietnam, have collectively signed the Agreement on the ASEAN Harmonized Cosmetic Regulatory Scheme (AHCRs) during the 35th ASEAN Economic Ministerial Meetings in Phnom Penh, Cambodia, on 2nd September 2003. The essence of the Agreement is the harmonization of rules and regulations which regulate cosmetics in ASEAN, whereby rules and regulations are to be revised for the pre-market product notification to be adopted. ASEAN Member States must comply with the ASEAN Cosmetic Directive starting from 1st January 2008 onwards in order to reduce the pre-market regulation and focus more on post-market surveillance, with a view to removing non-tariff barriers to trade by harmonizing the rules and regulations in relation to cosmetics in ASEAN for the benefit of promoting and facilitating trade and ensuring faster importation and exportation of cosmetics without compromising their safety according to international standards and ensuring consistency within the ASEAN Member Countries. For the past years the ASEAN Cosmetic Scientific Body (ACSB) and ASEAN Cosmetic Committee (ACC) have held a meeting twice a year since 2003, roughly in June and December of every year, where ASEAN Member Countries would take turns hosting the meetings.
As for Thailand, we have undertaken to develop several basic infrastructures, from cosmetic notification through electronic-submission system to capacity building of officials as well as entrepreneurs through different activities, such as training projects and government personnel and entrepreneur seminars, with a view to preparing them for the harmonization and changes of cosmetic-related rules and regulations in accordance with ASEAN. For the inspections of manufacturing sites of cosmetics and the surveillance of products in the market, the quality management system under ASEAN Cosmetic Good Manufacturing Practice (GMP) and Post-Market Surveillance (PMS) under ASEAN’s provisions on cosmetics have been adopted and applied to the context of Thailand. Similarly, entrepreneurs have been promoted and supported in their ability to compete both national and international levels.
1.2 ASEAN Agreement on Medical Device Directive (AMDD)
ACCSQ - MDPWG (ASEAN Consultative Committee for Standards and Quality - Medical Device Product Working Group) is a working group responsible for the negotiation and preparation of agreements between ASEAN Member States regarding rules and regulations on medical devices. The working group meets regularly and has appointed the Medical Device Technical Committee consisting of government representatives of ASEAN Member States. The Committee was tasked with the preparation of ASEAN Agreement on Medical Device Directive (AMDD), the first draft of which was completed in 2010, which went through several revisions before reaching the final draft in 2011, with 24 Articles and 8 Annexes, which then entered the national consultation process (National consultation - Final draft AMDD to be endorsed by the MDPWG) and was signed on 21st November 2014.
ASEAN Member States must ratify the AMDD to the ASEAN Secretariat in order for the AMDD to remain in effect in September 2021. So far, 9 Member States have ratified the agreement. Thailand has ratified the AMDD since 19th January 2021. The only remaining country which has not yet ratified the AMDD is Brunei Darussalam.
1.3 ASEAN Sectoral Mutual Recognition Arrangement (MRA) For Good Manufacturing Practice (GMP) Inspection of Manufacturers of Medicinal Products
ASEAN Member States signed the MRA on 10th April 2009. The MRA is designed to promote the recognition of GMP inspection reports of manufacturers of medicinal products sold in ASEAN which meet the international quality standards. At the moment, the Protocol to Amend the ASEAN Sectoral Mutual Recognition Arrangement for Good Manufacturing Practice (GMP) Inspection of Manufacturers of Medicinal Products is being prepared with a view to expanding the scope of the ASEAN MRA on GMP to cover Active Pharmaceutical Ingredients (APIs) and biopharmaceuticals. The Protocol was endorsed by the Pharmaceutical Product Working Group (PPWG) on 2nd February 2022, and by the ASEAN Consultative Committee for Standards and Quality (ACCSQ) on 14th February 2022. It was then endorsed by the Senior Economic Officials Meeting (SEOM) on 16th March 2022 and is aimed to be signed by the ASEAN Economic Ministers (AEM) in 2023.
1.4 ASEAN Mutual Recognition Arrangement for Bioequivalence Study Reports of Generic Medicinal Products (ASEAN MRA BE)
ASEAN initiated the ASEAN Mutual Recognition Arrangement for Bioequivalence Study Reports of Generic Medicinal Products (ASEAN MRA BE) with a view to enabling the mutual recognition of bioequivalence study reports issued by ASEAN listed bioequivalence study centers located in the territory of Member States. The MRA has entered into force since its signature on 2nd November 2017. Member States must comply with the MRA within 5 years after the MRA comes into force.
1.5 ASEAN Mutual Recognition Arrangement on Inspection and Certification System on Food Hygiene for Prepared Foodstuff Products
The MRA was prepared by the Prepared Foodstuff Product Working Group as a guideline for operations regarding food control system, food labeling, food hygiene, and food inspection and certification system, and to serve as a mutual guidance for food safety regulation among ASEAN Member States. The MRA is also consistent with the international standards, namely Codex. The MRA was signed by ASEAN Member States on 27th April 2018.
1.6 ASEAN Common Technical Dossier (ACTD) and ASEAN Common Technical Requirement (ACTR)
THE ASEAN Common Technical Dossier (ACTD) and the ASEAN Common Technical Requirement (ACTR) were prepared by the Pharmaceutical Product Working Group (PPWG). For more information, please visit https://asean.org/our-communities/economic-community/
standard-and-conformance/.
2. ASIAN Harmonization Working Party (AHWP)
Asian Harmonization Working Party (AHWP) is a non-profit organization established between 1996 - 1997 and consists of a group of members connected to the regulation of medical devices, both from the public sector and the private sector in the Asia Pacific region. The organization’s main objective is to study, review and provide opinion and recommendation regarding the regulation of medical devices to ensure its consistency among Asian countries and with other regions by cooperating with groups of international cooperation organizations such as International Medical Device Regulatory Forum (IMDRF), Asia-Pacific Economic Cooperation (APEC), and other agencies relating to the standards guideline development for the regulation of medical devices. Currently, the AHWP has 26 member countries from all over Asian, South American and African regions, and holds a general meeting every year. The Food and Drug Administration had the honor of hosting the 20th AHWP Annual Meeting on 2nd - 6th November 2015 at Dusit Thani Bangkok Hotel, Bangkok.
3. South-East Asia Regulatory Network (SEARN)
South-East Asia Regulatory Network (SEARN) is a network under the World Health Organization - South-East Asia Region (WHO-SEARO), established on 26th November 2016, with its main objective being the development and strengthening of collaboration between networks of national regulatory agencies within Southeast Asia and South Asia. The SEARN’s first meeting was held on 11th - 12th April 2017 in New Delhi, India. It consists of 11 member countries, being Thailand, the People’s Republic of Bangladesh, the Republic of India, the Republic of Indonesia, the Democratic People’s Republic of Korea, the Republic of the Union of Myanmar, the Kingdom of Bhutan, the Republic of Maldives, the Federal Democratic Republic of Nepal, the Democratic Republic of Timor-Leste, and the Democratic Socialist Republic of Sri Lanka. Thailand is one of its permanent members alongside the Republic of India and the Republic of Indonesia. The SEARN’s work is carried out through its working groups, which comprise representatives of its member countries, and puts emphasis on cooperation in 5 different areas: (1) Quality; (2) Regulatory Strengthening; (3) Vigilance; (4) Information; and (5) Medical Device.
4. The Pharmaceutical Inspection Co-operation Scheme (PIC/S)
The Pharmaceutical Inspection Co-operation Scheme (PIC/S) is an international-level organization with government agencies responsible for pharmaceutical regulation from different countries as members. The PIC/S’ role is to issue rules on Good Manufacturing Practice (GMP) for the pharmaceutical manufacturing, and to inspect and approve national regulatory authorities on pharmaceutics of the member countries to ensure their compliance with the prescribed standards and criteria. Many countries from Europe, Africa, America and Asia have joined the PIC/S as members, and the number of countries being interested in joining is increasing. Joining the PIC/S can benefit a country as it helps promote the country’s pharmaceutical industry in terms of exportation of products to other member countries, as this would be more convenient after joining. Consequently, the country’s value of export is increased, which is another benefit to the country’s economy.
The Food and Drug Administration foresees the significance of the promotion of pharmaceutical business, which would help achieve the national policy goals on drugs in terms of pharmaceutical industry development. As a result, the Food and Drug Administration has constantly promoted these businesses in order to drive forward these strategic goals together with other relevant sectors. Consequently, Thailand has joined the PIC/S since 1st August 2016, and has issued the Notification of the Ministry of Public Health Re: Prescription of Details Relating to Rules and Procedures on the Manufacturing of Modern Medicines under the Law on Drugs, B.E. 2554 (2011), dated 14th April, B.E. 2554 (2011), in compliance with the PIC/S GMP, urging pharmaceutical entrepreneurs in the country to improve their manufacturers in compliance with the standards
